Dear Dr. Zoomie – your last posting discussed how gas-filled detectors work, but I’ve got a different type. I think it’s called a sodium iodide detector. Can you tell me how this works and when I should use it? Thanks!
You’ve got one of my favorite detectors (and yes, I know that this raises me way up on Geek sale)! There are two fundamental families of detectors – the gas-filled detectors I wrote about the last time, and the scintillation detectors, of which the sodium iodide (abbreviated NaI) is one. The vast majority of radiation detectors out there – and virtually every detector used in general situations – fall into one of these two families. So since I wrote about the gas-filled detectors last time, this is a good chance to write about the other major family. Here’s how the scintillation-type detectors work.
First, an important point. Most scintillation detectors are only sensitive to one type of radiation. So NaI detectors will pick up gamma radiation, but not alpha or beta, zinc sulfide (ZnS) will only pick up alpha radiation, and so forth. In actuality, there might be some sensitivity to other radiations – NaI, for example, will sometimes pick up high-energy betas – but you should only use a detector for the type of radiation it’s designed to pick up. Now, with that out of the way, on to how the things work!
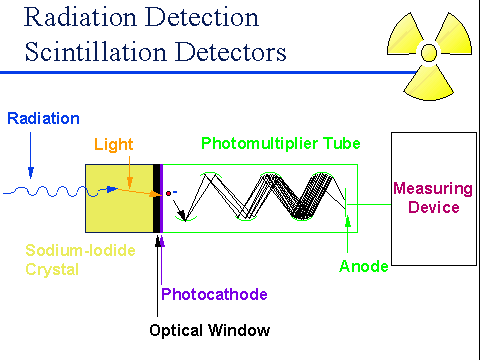
The basic principle is the same for every scintillation-type detector: when radiation strikes the scintillator it causes it to give off photons of visible light (that’s the scintillation part). These photons pass through the crystal and they strike a thin metal foil called a photocathode – when this happens the light enters the second part of the detector, called a photo-multiplier tube (PMT). When the photon hits the photocathode it causes an electron to be ejected from the photocathode. Just past the photocathode there is a set of metal cups, each with a voltage applied to it (typically several hundred to a thousand volts) – the electron is accelerated by this voltage to a high energy and it strikes the cup with enough energy that it knocks loose a number of other electrons. Each of them, in turn, is accelerated towards the next metal cup, where each of the “new” electrons knocks loose a number of additional ones – by the end of the PMT the initial signal has been multiplied by a factor of a million or so. From there, it’s up to the instrument manufacturer to figure out how they’re going to use the light that’s emitted.
As one example, every time a gamma hits the crystal it starts this whole process that culminates in a pulse of electrons arriving at the far end of the detector. The simplest way to deal with this is to simply count the pulses of electrons as they arrive at a counting circuit – this is a great way to measure contamination (which we normally record in terms of counts per minute or counts per second). We can also use this mode to measure radiation dose rates, but only by assuming that every count carries with it a specific amount of energy. If you remember the posting on gas-filled detectors, this is the same way that Geiger counters work and it’s one of the reasons that Geiger counters aren’t always accurate for measuring radiation dose rates. We’ll get back to that in a moment. Oh – one other thing to be careful about with an NaI detector is that the larger crystals (say, 2”x2”) can be sensitive to drastic temperature changes – they can thermally stress the crystal, eventually breaking it. So if you’re working outside on a blazing hot (or bitingly cold) day, you probably want to leave the crystal outside, rather than bring it into a much colder (or warmer) office a few times a day.
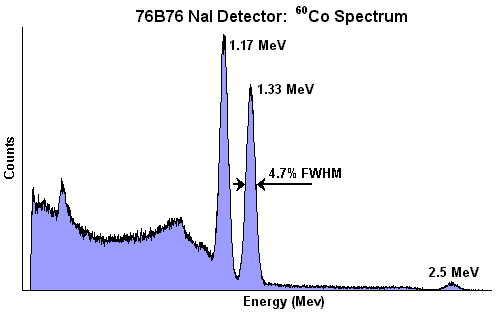
Something else that we can use NaI detectors for is identifying specific radionuclides by measuring the energy of each individual gamma that enters the crystal – this process is called gamma spectroscopy, or can also be called multi-channel analysis (and the instrument set up for this purpose is called a gamma spectroscopy device, or a multi-channel analyzer – abbreviated MCA). The basic principle behind gamma spectroscopy is that every gamma-emitting radionuclide emits a gamma ray (or a few gammas) with very specific energies – like a fingerprint – and if we can identify the gamma energies precisely enough then we can identify the radionuclide(s) present. For example, cesium-137 (Cs-137) gives off a gamma with an energy of 662 thousand electron volts (abbreviated keV) – if we analyze a gamma ray spectrum and find a peak with an energy of 662 keV then we know that Cs-137 is present. Along the same lines, seeing twin gamma peaks at about 1.1 and 1.3 million electron volts (MeV) tells us that we’ve found cobalt-60 (Co-60).
The problem is in figuring out how much energy is in each gamma photon – luckily we can do this with a scintillation detector. When a gamma ray interacts with the NaI crystal it deposits energy – this energy is what causes the photons to be given off. Not only that, but a predictable number of photons are emitted depending on the energy deposited – in a sodium iodide crystal, depositing 1 MeV of energy will cause about 42,000 scintillation photons to be emitted. We know the number of photons that it takes to eject a single electron from the photocathode, and we know the amount of amplification (and the size of the electrical pulse) for each electron ejected. So if we can measure the size of the output pulse then we know how much energy was deposited in the crystal – and we can know what radionuclide emitted the gamma that we just detected.
So these are two uses of NaI detectors – measuring contamination levels (in counts per minute or counts per second) and identifying radionuclides by measuring gamma energies – now for a third, measuring radiation dose rate.
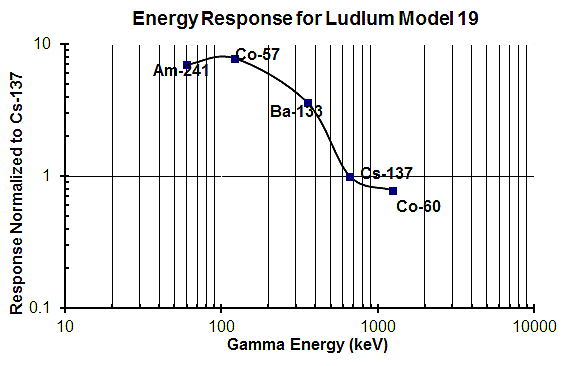
Radiation dose rate is a measure of the amount of energy deposited in an object. You’d think that this would be fairly simple with an NaI detector since the number of electrons coming out of the detector is proportional to the energy deposited in the crystal. But that’s not how most manufacturers handle things – most of the time they simply count the pulse rate and, as with a Geiger counter, assume that they are all coming from Cs-137. So, for example, the manufacturer might determine that 175 counts per second are equivalent to a dose rate of 1 mR/hr. This means that NaI detectors set up to measure dose rate this way have the same limitations as a Geiger counter – unless you are measuring exactly what they were calibrated with the dose rates you measure are going to be wrong. Thus, if you’re using a scintillation detector to measure radiation dose rate you need to make sure that either you’re measuring the same nuclide to which it was calibrated or you need to have a set of correction factors that will let you convert the meter reading to the correct dose rate (assuming that you know the nuclide that’s actually present). This graphic, which is for a sodium iodide scintillation detector, show that the meter will show only half the actual dose rate if the radiation is from a Co-60 source, but will over-respond by a factor of 6 or 7 if the radiation is coming from the Am-241 in, say, a box of smoke detectors.
OK – so that covers how the NaI detector works and what it can be used for (again, to measure gamma contamination, for nuclide identification, or – with caveats – to measure radiation dose rate). Now a little on other types of scintillators.
Zinc sulfide (ZnS) is used to measure alpha contamination. The ZnS crystals are razor-thin – only about as thick as a single human hair (give or take a little). But since alpha particles can’t penetrate very far into any materials the crystals don’t need to be any thicker than this. For a number of reasons we don’t worry about dose rate from alpha radiation, so the only thing we need to measure is count rate – a fairly simple matter of counting pulses. The biggest problem with ZnS detectors is that they can be fragile (remember how thin the crystals are). It can also take a long time to do a proper alpha contamination survey since alphas are so easily shielded and have such a short range in air. But for alpha counting, ZnS is about as good as it gets, in spite of its limitations.
Finally, there are also beta scintillators. Liquid scintillation counting is normally performed in the laboratory using fairly expensive (and large) machines – chances are that you won’t have to use one of these unless you work in a laboratory. There are also beta scintillation crystals that you use the same way you use an NaI detector – these tend to be made of plastic (called organic scintillators). While not as fragile as ZnS or NaI, the photo-multiplier tube is the same in all of these detectors and is not very sturdy – no matter what kind of scintillation detector you’re using, you need to treat it gently.
So to sum all of this up, scintillation detectors have their limitations, but they are essential pieces of equipment for just about anyone making radiation measurements. The big things are to treat them gently to keep from breaking them, and to use them properly and within their limitations.